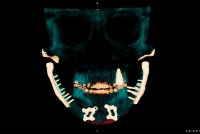
FDA Said It Never Inspected Dental Lab That Made Controversial AGGA Device
Johns Dental Laboratories stopped making the Anterior Growth Guidance Appliance last year after a ƒ¢πΩ”∞‘∫ Health News-CBS News investigation into allegations of patient harm. The company had ‚Äúnever‚Äù reported any complaints about its products to the FDA, according to the agency.
The Horrors of TMJ: Chronic Pain, Metal Jaws, and Futile Treatments
TMJ disorders affect as many as 1 in 10 Americans and yet remain poorly understood and ineffectively treated. Many common treatments used by dentists lack scientific evidence.
‘AGGA’ Inventor Testifies His Dental Device Was Not Meant for TMJ or Sleep Apnea
The FDA and Department of Justice are investigating the Anterior Growth Guidance Appliance, or “AGGA.” TMJ and sleep apnea patients have filed lawsuits alleging the device harmed them. Its inventor now says the AGGA was never meant for these ailments.
Patients Expected Profemur Artificial Hips to Last. Then They Snapped in Half.
The FDA and the manufacturer were alerted to Profemur titanium hips breaking inside U.S. patients as of 2005. It took 15 years to recall the devices. Many fractures could have been avoided.
After Backlash, Feds Cancel Plan That Risked Limiting Breast Reconstruction Options
The Centers for Medicare & Medicaid Services backed off from a plan that could have curtailed access to a type of reconstructive surgery known as DIEP flap. Breast cancer patient advocates are relieved.
How a Medical Recoding May Limit Cancer Patients’ Options for Breast Reconstruction
The federal government’s arcane process for medical coding is influencing which reconstructive surgery options are available, creating anxiety for breast cancer patients.
Feds Launch Criminal Investigation Into ‘AGGA’ Dental Device and Its Inventor
ƒ¢πΩ”∞‘∫ Health News and CBS News recently reported that multiple lawsuits allege the device has led to grievous injuries to patients‚Äô mouths, resulting in loss of teeth.
FDA Evaluates ‘Safety Concerns’ Over Dental Devices Featured in KHN-CBS Investigation
A KHN and CBS News investigation found that a dental appliance called the AGGA has been used by more than 10,000 patients, and multiple lawsuits allege it has caused grievous harm to patients.
FDA Looks Into Dental Device After KHN-CBS News Investigation of Patient Harm
The FDA’s interest in the AGGA dental device follows a KHN-CBS News investigation, according to a former agency official.
A los pocos meses de usar AGGA, una paciente dijo que sus dientes estaban tan flojos que podía sentir cómo se movían cuando se untaba crema hidratante en las mejillas. Besar a su novio le resultaba incómodo.
This Dental Device Was Sold to Fix Patients’ Jaws. Lawsuits Claim It Wrecked Their Teeth.
A dental device called AGGA has been used on about 10,000 patients without FDA approval or proof that it works. In lawsuits, patients report irreparable harm. The AGGA’s inventor and manufacturer have denied all liability in court.